what’s in the works
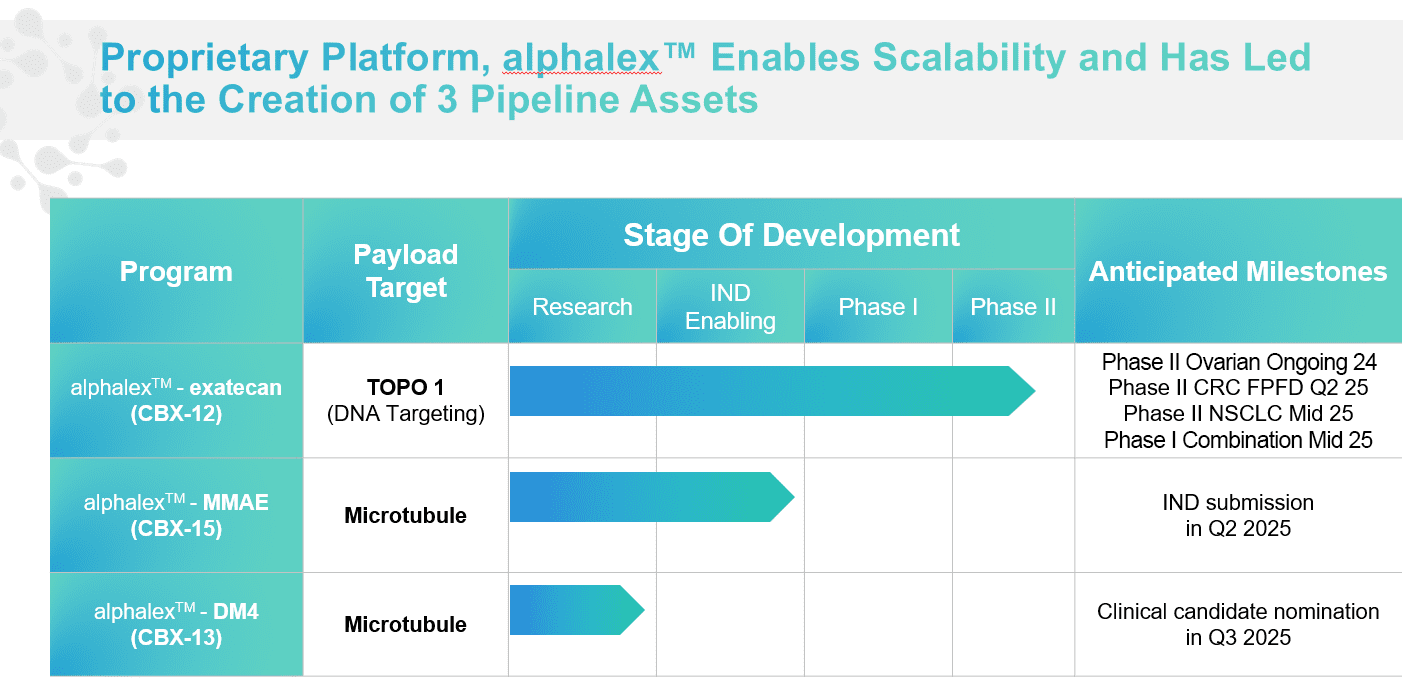
CBX-12 is an alphalex conjugate that includes the highly potent topoisomerase I inhibitor exatecan. CBX-12 was selected based on its ground-breaking efficacy and safety profile demonstrated in preclinical studies. CBX-12 has potential synergy with PD-1 inhibitors and other immuno-oncology drugs.
development focus: toxin conjugates
Highly potent anti-cancer agents that are unable to be administered as free drug are targeted to tumors with alphalex, creating novel agents that are effective across tumor types.
presentations & publications
Learn more about the science of alphalex and how it powers our work at Cybrexa.

Characterization of Antigen Agnostic Tumor Selective Delivery and Immunomodulatory Activities of Auristatin and Maytansinoid alphalex™ Peptide-Drug Conjugates
Download
669P: CBX-12-101: Final Results of a Phase 1 Study of CBX-12, a Peptide Drug Conjugate (PDC) in Patients (pts) with Advanced or Metastatic Solid Tumors.
Download
Victoria ADC Meeting
Download
Selection of Tubulin & Topl Inhibitor Payloads for Improved Tumor Specific Delivery by Peptide Drug Conjugate
Download
CBX-12-101: A First-in-Human Study of CBX-12, an Alphalex™ Peptide Drug Conjugate (PDC) in Patients with Advanced or Metastatic Solid Tumors
Download
Evaluation of antigen-agnostic anti-tumor activity and immunological memory induced by CBX-15 (alphalex™-MMAE) in the rat syngeneic breast cancer model
Download
TOP1-DNA trapping by exatecan and combination therapy with ATR inhibitor
Download
Antigen-independent tumor targeting by CBX-12 (alphalex™-exatecan) induces long-term antitumor immunity
Download
CBX-12-101: A first-in-human study of CBX-12, an alphalex™ peptide drug conjugate (PDC) in patients with advanced or metastatic solid tumors
Download
Development of an alphalex™-auristatin low pH targeting conjugate for the treatment of solid tumors
Download
Tumor-selective, antigen-independent delivery of a pH sensitive peptide-topoisomerase inhibitor conjugate suppresses tumor growth without systemic toxicity
Download
Development of alphalex™-auristatin low pH targeting conjugates for the treatment of solid tumors
Download
CBX-12 (alphalex™-exatecan) sensitizes tumors to immune checkpoint blockade in an antigen agnostic manner by immune activation
Download
Development of alphalex™-toxin low pH targeting conjugates for the treatment of solid tumors
Download
CBX-12: A low pH targeting alphalex™-exatecan conjugate for the treatment of solid tumors
Download
Targeting solid tumor acidic microenvironment with alphalex™ PARP inhibitor
Download
Development of tumor-targeted PARP inhibitors for the treatment of solid cancers
Download
MicroRNA silencing for cancer therapy targeted to the tumour microenvironment
Download
